CCHD Screening with Pulse Oximetry
Reliable, Clinically-Proven Screening for Critical Congenital Heart Disease (CCHD) with Masimo SET®
Home / Newborn Care / CCHD Screening
CCHD Screening
Critical Congenital Heart Disease
Critical Congenital Heart Disease
Congenital heart defects (CHDs) are the most common birth-related malformation.1 Critical CHDs (CCHDs) are life threatening and account for nearly 25% of all CHDs.2 Babies sent home with an undetected heart defect are at risk of serious complications within the first few days or weeks of life, requiring emergency care,3 and even at risk of dying.
CCHD Screening with Pulse Oximetry
CCHD Screening with Pulse Oximetry
Traditionally, following birth, newborns were observed for evidence of CHDs by physical assessment and monitoring for common symptoms.1 Today, studies show that these methods alone can be unreliable and may fail to detect up to 36% of infants with CCHD before discharge.4,5 Adding screening with pulse oximetry can help diagnose CCHD before an infant becomes symptomatic.6
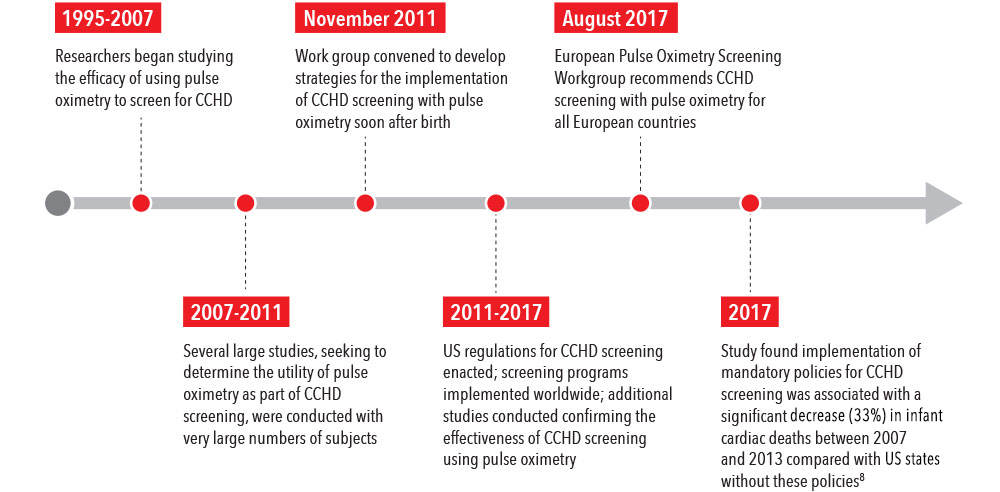
In 2011, following numerous studies observing the utility of pulse oximetry in screening for CCHD, a work group was convened to develop strategies for the implementation of safe, effective, and efficient CCHD screening with pulse oximetry.7 The work group found sufficient evidence to recommend the use of pulse oximetry to screen for CCHD, and further recommended that screening be performed with motion-tolerant pulse oximeters that report functional oxygen saturation (SpO2) and have been validated in low-perfusion conditions.7
In a recent study, researchers found a 33% decrease in infant cardiac deaths after US statewide implementation of mandatory policies for CCHD screening, compared with prior periods and states without policies, as well as a 21% decrease in early infant deaths from other/unspecified cardiac causes.8
Why is it Important to use Masimo SET® Pulse Oximetry for CCHD Screening?
Why is it Important to use Masimo SET® Pulse Oximetry for CCHD Screening?
Masimo’s breakthrough Signal Extraction Technology® (SET®) overcomes the limitations of conventional pulse oximetry with the ability to measure through motion and low perfusion. Masimo SET® was also the first pulse oximetry technology to receive FDA 510(k) clearance in the labeling, in 2012, to screen newborn patients for CCHD.9
Improved Critical Congenital Heart Disease Screening Sensitivity vs. Clinical Assessment Alone5
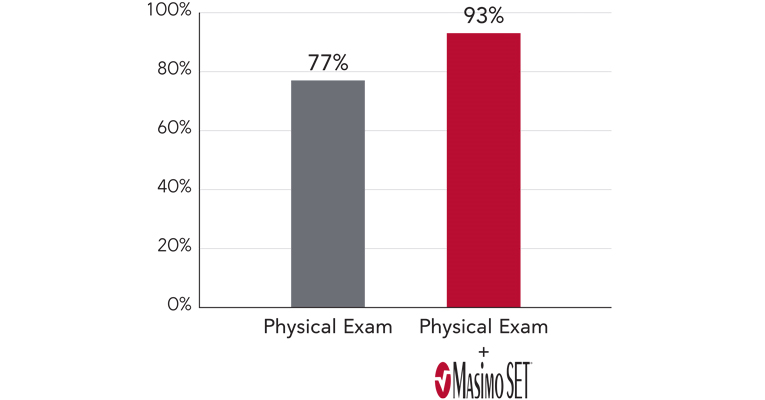
When combined with clinical assessment, Masimo SET® improved CCHD screening sensitivity to 93%5
There have been multiple large published CCHD screening studies that exclusively used Masimo SET® pulse oximeters and sensors, two of which (59,876 subjects) were the basis for the CCHD workgroup recommendation for CCHD screening protocols.4,7,12 Cumulatively, six of these studies represent over 284,800 infants4,5,10-13 and include the largest CCHD screening study to date, with over 122,738 subjects.5
These six notable studies, all using Masimo SET®, concluded that pulse oximetry, in conjunction with clinical assessment, improved screening sensitivity compared to routine physical exam alone.4,5,10-13

In addition, Masimo SET® measures perfusion index (Pi). Incorporating perfusion index into screening has been shown to increase sensitivity to the detection of CCHD in infants with pathologically low perfusion.4 In a study of 10,009 infants, when Pi was added to CCHD screening, screening revealed abnormalities in 100% of all newborns who had left heart obstructive disease (LHOD).10
How to Conduct CCHD Screening with Pulse Oximetry
How to Conduct CCHD Screening with Pulse Oximetry
CCHD screening using pulse oximetry can be easily implemented following the work group’s recommended protocols.
After 24 hours of life, or as close to discharge as possible, screening is conducted by taking a preductal SpO2 measurement on the right hand followed by a postductal SpO2 measurement on either foot. Based on the measurement results, the protocol will identify a positive or negative screening result, or recommend that repeat screening be conducted.7
The Work Group’s Recommended CCHD Screening Protocol
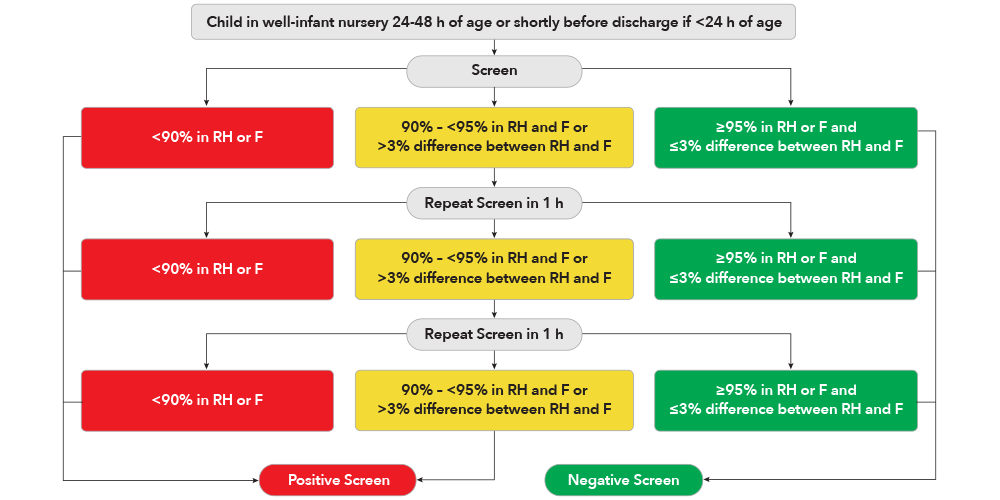
To improve the early detection of CCHD, the US Secretary of Health and Human Services (HHS) recommended that CCHD screening be added to the uniform newborn screening panel.14 One of the well accepted methods for screening newborns for CCHD is a protocol endorsed by the Secretary’s Advisory Committee on Heritable Disorders in Newborns and Children (SACHDNC), American Academy of Pediatrics (AAP), the American College of Cardiology Foundation (ACCF) and the American Heart Association (AHA) and based on pulse oximetry studies by Granelli et. al.4,7 It involves measuring pre-ductal (right hand, RH) and post ductal (foot, F) functional oxygen saturation (SpO2) levels in newborns after 24 hours of age.
Masimo Solutions for CCHD Screening with Pulse Oximetry
Masimo Solutions for CCHD Screening with Pulse Oximetry
Masimo offers a variety of pulse oximeters and sensors equipped with SET® technology for CCHD screening.
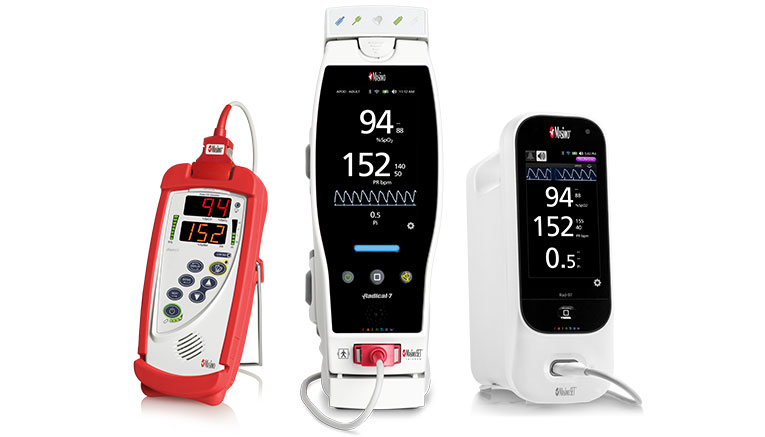
Radical-7 and Rad-97 Pulse CO-Oximeters can be mounted on a portable roll stand to easily move between rooms and include advanced connectivity solutions that enable automated documentation of screening measurements in the hospital’s EMR. Masimo’s roll stand solutions feature wheels that glide smoothly and quietly, as well as a basket to store sensors and other supplies and a bracket for disinfecting wipes.
A Rad-5® handheld pulse oximeter is also available, allowing quick screening and easy transport between rooms.
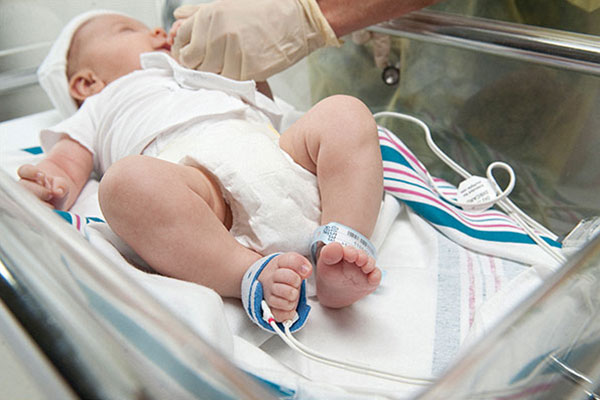
Multisite Y-I reusable sensors with soft foam wraps, used in multiple prominent CCHD screening studies,4,5,10-13 enable quick, comfortable application and removal, without disturbing infants.

RD SET™ Neo single-patient-use, wrap-around style sensors feature a flat, lightweight cable that lies comfortably on the patient's hand or foot.
Learn More About CCHD Screening with Masimo SET®
Learn More About CCHD Screening with Masimo SET®
References:
- 1.
Ewer AK, et al. NIHR Health Technology Assessment Programme: Executive Summaries. 2012.
- 2.
https://www.cdc.gov/ncbddd/heartdefects/data.html
- 3.
https://www.cdc.gov/ncbddd/heartdefects/cchd-facts.html
- 4.
de-Wahl Granelli A et al. BMJ. 2009;Jan 8;338.
- 5.
Zhao et al. Lancet. 2014 Aug 30;384(9945):747-54.
- 6.
www.thelancet.com/child-adolescent. Published online August 30, 2017 http://dx.doi.org/10.1016/S2352-4642(17)30066-4
- 7.
Kemper AR et al. Pediatrics. 2011 Nov;128(5):e1259-67.
- 8.
Rahi Abouk et al. JAMA. 2017;318(21):2111-2118. doi:10.1001/jama.2017.17627.
- 9.
Masimo 510(k) Summary - K120657
- 10.
de-Wahl Granelli A et al. Acta Paediatr. 2007 Oct;96(10):1455-9.
- 11.
Meberg A et al. Pediatr. 2008 Jun;152(6):761-5.
- 12.
Ewer AK et al. Lancet. 2011 Aug 27;378(9793):785-94.
- 13.
Schena F et al. J Pediatr. 2017;183:74-79.
- 14.
Secretary’s Advisory Committee on Heritable Disorders in Newborns and Children. HHS Secretary adopts recommendation to add critical congenital heart disease to the Recommended Uniform Screening Panel. September 21, 2011. Washington, DC: US Department of Health and Human Services.
RESOURCES
Eve has obtained CE Marking. Not available in the U.S.
For professional use. See instructions for use for full prescribing information, including indications, contraindications, warnings, and precautions.
PLCO-002052/PLM-11369A-0718