Technologies
Cardiac Output (CO) Monitoring
Cardiac output (CO) is the amount of blood that is ejected from the heart per minute. Monitoring the components of CO allows clinicians to assess if there is a sufficient volume of blood in the body to transport oxygen. Monitoring CO can help clinicians manage fluids, monitor therapeutic interventions, and improve patient outcomes.1
CO monitoring, along with other advanced hemodynamic parameters, is available through the Masimo LiDCO® Hemodynamic Monitoring System.
Fluid Optimization
Fluid administration is one of the most common interventions made to increase CO. However, fluid administration should be balanced to avoid both hypovolemia and hypervolemia, which have both been associated with negative outcomes.2
Components of Oxygen Delivery
Oxygen delivery (DO2) is the amount of oxygen delivered to the tissues, calculated as the product of CO and oxygen content (CaO2).3
CO is calculated by multiplying the stroke volume (SV) by the patient’s heart rate (HR). SV is the amount of blood pumped by the left ventricle of the heart in one contraction.3
DO2 and CaO2 are influenced by the patient's Oxygen Saturation (SaO2) and Hemoglobin (Hb).3
Normal Hemodynamic Parameters4-8
| Parameter | Equation | Normal Range |
| Oxygen Delivery (DO2) | CaO2 x CO x 10 | 950-1150 mL/min |
|---|---|---|
| Cardiac Output (CO) | HR x SV/1000 | 4.0 – 8.0 L/min |
| Oxygen Content (CaO2) | (1.38 x Hgb x SaO2) + (0.0031 x PaO2) | 17 – 20 mL/dL |
| Stroke Volume (SV) | N/A | 60 – 100 mL/beat |
| Oxygen Saturation (SaO2) | N/A | 95 – 100% |
| Total Hemoglobin (Hb) | N/A | Men: 13.8-17.2 g/dL Women: 12.1-15.1 g/dL |
| Heart Rate (HR) | N/A | Range varies based on patient status (resting vs. active, age, etc.) |
PulseCO™ Algorithm Technology Overview
The PulseCO™ algorithm provides continuous beat-to-beat CO and SV by analyzing a blood pressure waveform. The algorithm is based on physics and physiological principles and focuses on pulse power analysis rather than waveform shape or contour. Unlike other arterial pressure algorithms, PulseCO is not based on statistics and assumptions about vascular compliance, nor on the detection of the dicrotic notch, which is often a challenge with peripheral arterial signals. As a result, the PulseCO algorithm avoids the limitations of other pulse pressure or contour-based hemodynamic monitoring technologies.
The current gold standard in hemodynamic monitoring, although not as commonly used due to its invasiveness, is the pulmonary artery catheter (PAC). The PulseCO algorithm has been validated against the PAC demonstrating a good agreement between the two methods.9
PulseCO Clinical Evidence
Reductions in 30-Day and 180-Day Mortality
In a study comparing the outcomes of 1200 major abdominal surgery patients, researchers found that, following the implementation of a program including LiDCO Monitoring with PulseCO technology, there was a significant decrease in mortality at 30 days (from 21.8 to 15.5%) and 180 days (from 29.5 to 22.2%).10
Reductions in Postoperative Complications and Costs
In a randomized, controlled trial of 734 patients undergoing major gastrointestinal surgery, researchers found hemodynamic optimization with LiDCO Monitoring with PulseCO technology led to more than 20% reduction in postoperative complications and, as a result, patients monitored with LiDCO Monitoring with PulseCO technology were on average $530 less expensive to treat over 6 months than control patients who were not monitored.1, 11
Masimo LiDCO Product Portfolio

Masimo LiDCO™
Hemodynamic Monitoring with Root®
Beat-to-beat advanced hemodynamic monitoring to support clinical decision making in perioperative care settings
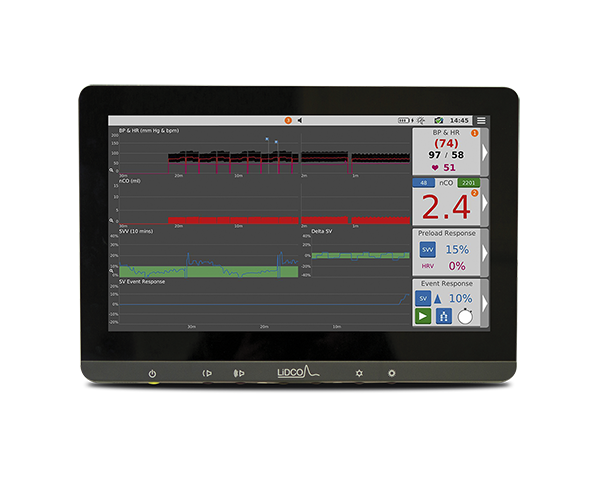
LiDCO® Rapid
Hemodynamic Monitoring System
Beat-to-beat advanced hemodynamic monitoring to support informed decision-making in high-acuity care areas
RESOURCES
References:
Pearse R et al. JAMA 2014; 311(21):2181-90.
Bellamy MC. Br J Anaesth. 2006 Dec;97(6):755-7.
Miller's Anesthesia, 8th Edition, Vol. 2
Burns, S. M., & Delgado, S. A. (2019). AACN essentials of critical care nursing (4th ed.). New York, NY: McGraw-Hill.
Diepenbrock, N. H. (2015). Quick reference to critical care (5th ed.). Philadelphia, PA: Wolters Kluwer.
Jones, J., & Fix, B. (2015). Critical care notes: Clinical pocket guide (2nd ed.). Philadelphia, PA: FA Davis.
Urden, L. D., Stacy, K. M., & Lough, M. E. (2020). Priorities in critical care nursing (8th ed.). St. Louis, MO: Elsevier.
World Health Organization Global Database on Anaemia. 2008.
Costa et al. Intens Care Med. 2007. DOI 10.1007/s00134-007-0878-6 P1.8
Tengberg LT et al. Br J Surg 2017; 104:463-471.
Sadique Z, et al. Perioper Med (Lond). 2015 Dec 14;4:13.
For professional use. See instructions for use for full prescribing information including indications, contraindications, warnings and precautions. Caution: Federal (USA) law restricts this device to sale by or on the order of a physician.
PLCO-007995/PLM-13197C-1125