 Masimo Signal Extraction Technology® (SET®)
Masimo Signal Extraction Technology® (SET®)
In 1989, two young engineers asked themselves why pulse oximetry wouldn't work during patient motion and low perfusion–and by doing so, set a new course that created a revolution in patient monitoring.
Home / SET Pulse Oximetry
SET Pulse Oximetry
Masimo Signal Extraction Technology (SET®)
Masimo Signal Extraction Technology (SET®)
Overcoming the Limitations of Conventional Pulse Oximetry
Since its inception, pulse oximetry was plagued by unreliability when it was needed most–during patient motion and low perfusion. The industry had given up and considered the problem "unsolvable." Clinicians were forced to live with the results–excessive false alarms, delayed notification due to long averaging times, inaccurate data, and an inability to obtain data on the most critical patients.
Conventional pulse oximetry works under the assumption that by looking at only the pulse and normalizing the pulsating signal over the non-pulsating signal, oxygen saturation (SpO2) can be measured without calibration. Although this was a big step forward in the evolution of pulse oximetry, it has one major flaw–it assumes the only pulsating component is arterial blood. Unfortunately for conventional pulse oximetry, venous blood moves every time the patient moves or breathes. This causes conventional pulse oximeters to display false low or high SpO2 and pulse rates–resulting in false alarms as high as 90% in ICUs and recovery rooms.1
The Choice of Many Clinicians in Leading Hospitals2
During challenging conditions of motion and low perfusion, clinicians at thousands of hospitals around the world count on Masimo SET® every day to help them care for patients. And while many leading hospitals have already integrated Masimo SET® pulse oximetry technology,2 more are converting every day.
These hospitals and clinicians use Masimo SET® to help them deliver patient care. With fewer false alarms,3 clinicians can focus on the patients who need the most attention. And with more timely detection of true events, clinicians can intervene earlier for better patient outcomes and improved patient safety.
Clinical Background
Clinical Background
To date, more than 100 studies have shown that Masimo SET® outperforms other pulse oximetry technologies, providing clinicians with unmatched sensitivity and specificity to make critical patient care decisions.4
Discovering Breakthrough Performance
When Joe Kiani and Mohamed Diab looked at the same pulse oximetry signal differently than anyone had before, they created new possibilities. By employing advanced signal processing techniques–including parallel engines and adaptive filters–they believed they could find the true arterial signal that would allow accurate monitoring of arterial oxygen saturation and pulse rate, even during the most challenging conditions. Signal Extraction Technology®, or Masimo SET®, assumes that both the arterial and venous blood can move and uses parallel signal processing engines–DST®, FST®, SST™, and MST™–to separate the arterial signal from sources of noise (including the venous signal) to measure SpO2 and pulse rate accurately, even during motion.
After six years of dedicated and focused research and development, Masimo SET® debuted in 1995 at the Society for Technology in Anesthesia and won the prestigious Excellence in Technology Innovation Award. Thereafter, skeptical clinicians around the world sought to compare Masimo SET® to the best pulse oximetry technologies other companies had to offer. But in study after study, the signal processing of Masimo SET®consistently resulted in significantly fewer false alarms and true alarm detection.
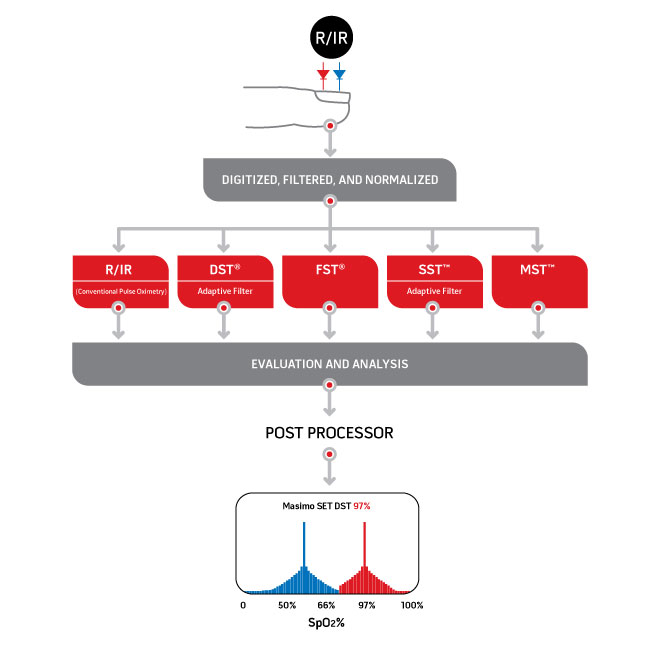
Conventional pulse oximetry uses the standard red over infrared algorithm to provide SpO2, while Masimo SET® uses that conventional algorithm but has added four other algorithms that all run in parallel. These algorithms allow the distinction between arterial and venous signal during motion and low perfusion by identifying and isolating the non-arterial and venous noise SpO2 (left peak shown in blue) from the true arterial SpO2 components (right peak shown in red) in the signal. The plot peak on the right is then chosen as the SpO2 value, since the physiologically higher SpO2 value within the measuring site will be arterial signal.
Alarm Reliability with Signal Extraction
Performance During Motion and Low Perfusion5*
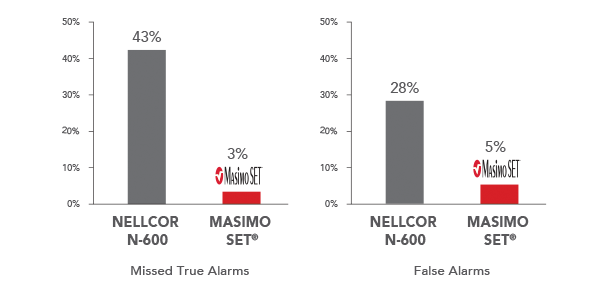
*Results shown are calculated by combining sensitivity and specificity outcomes of machine-generated and volunteer-generated motion. Investigators measured SpO2 in 10 subjects during motion and low perfusion conditions and calculated the false alarm rate (specificity) and true alarm rates (sensitivity).
Performance with Children7
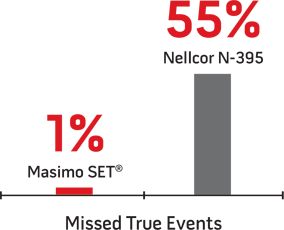
Researchers measured missed true desaturation events out of 75 true events in this study on 5 children undergoing evaluation for sleep-disordered breathing.
Performance with Infants8
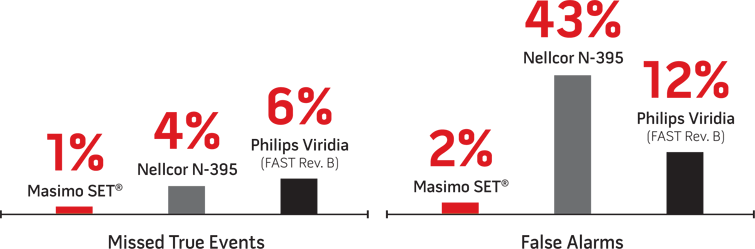
Pulse Oximetry ROC Curves3
Learn More About Masimo SET®
Learn More About Masimo SET®
References:
- 1.
Malviya S., Reynolds P.I., Voepel-Lewis T., Siewert M., Watson D., Tait A.R., Tremper K. Anesth Analg. 2000 Jun;90(6):1336-40.
- 2.
U.S. News & World Report, Best Hospitals 2015 Edition.
- 3.
Barker S.J. Anesth Analg. 2002 Oct;95(4):967-72.
- 4.
All studies available at http://www.masimo.com/home/clinical-evidence/clinical-evidence/.
- 5.
Shah N., Ragaswamy H.B., Govindugari K., Estanol L. J Clin Anesth. 2012 Aug;24(5):385-91.
- 6.
Barker S.J., Morgan S.E. Anesthesiology 2000; 93 (3A):A-549.
- 7.
Brouillette RT, Lavergne J, Leimanis A, Nixon GM, Laden S, McGregor CD. Differences in Pulse Oximetry Technology can Affect Detection of Sleep Disordered Breathing in Children. Anesth Analg. 2002; 94:S47-S53
- 8.
Hay WW, Rodden DJ, Collins SM, Melara DL, Hale KA, Fashaw LM. Reliability of conventional and new oximetry in neonatal patients. Journal of Perinatology. 2002; 22:360-266
RESOURCES
Video
PLCO-003841/PLM-11172A-0420





